Ecopipam Tablets to Study Tourettes's Disorder in Children, Adolescents and Adults
dialogue with the study sponsor, principal investigator, site staff, and participants, while ensuring compliance with confidentiality agreements, Good Clinical Practices, and both FDA and IRB regulations.
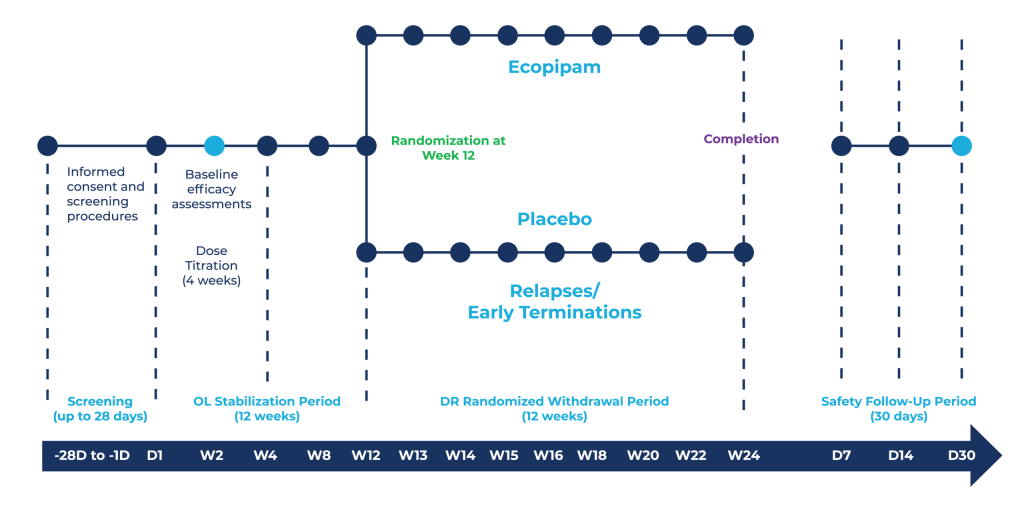
Visits consisted of completing a range of rating scales, assessments, and medical procedures administered by the PI and/or the site coordinator. My visit responsibilities included managing appointment structures, recording vitals, performing ECGs, answering any procedure or protocol related questions, and various other duties.

A multi-center, double-blind, placebo-controlled, randomized withdrawal study to evaluate the safety and maintenance of efficacy of ecopipam in children, adolescents, and adults with Tourette’s Disorder. This Phase 3 trial aimed to assess the safety and efficacy of the selective D1 dopamine receptor blocking agent, ecopipam, for the treatment of tics in children and adults with Tourette’s Syndrome.
Through targeted recruitment efforts the two Cortica sites surpassed program-wide enrollment goals. As a site coordinator I managed our daily operations from scheduling and running visits to data entry, queries, and audits. Throughout, I maintained continuous